NOTA TÉCNICA
doi: http://dx.doi.org/10.7705/biomedica.v35i2.2570
Author's contributions:
Andrea von Groll and Pedro Eduardo da Silva designed the study
Fernanda Abilleira and Clarice Brum performed the susceptibility tests
All authors contributed to the analysis of the data and the writing of the article
Recibido: 15/10/14; aceptado: 16/12/14
Introduction: Reports of Mycobacterium tuberculosis resistant to multiple drugs are increasing globally and laboratories are becoming increasingly aware of the need for drug susceptibility testing. In recent years, due to the long time required by conventional drug susceptibility testing, new approaches have been proposed for faster detection of drug resistance, such as the nitrate reductase assay, considered fast and inexpensive, making it a good diagnostic tool for low resource countries.
Objective: The present study proposed a fast direct colorimetric drug susceptibility testing method in a microplate format using solid medium.
Materials and methods: The diagnostic accuracy was evaluated by comparing the proportion method with the direct nitrate reductase assay in plates. Frozen sputum samples, known to be positive, were decontaminated and processed by Petroff method. The decontaminated suspension was used to perform direct nitrate reductase assay in 7H11 medium using 1 µ g/ml rifampicin (RIF), 0.2 µ g/ml isoniazid (INH), 2 µ g/ml ofloxacin (OFX), 6 µ g/ml kanamycin (KAN), 2 µ g/ml amikacin (AMK) and 10 µ g/ml capreomycin (CAP). Eighty-four samples were tested and the results for 69% of them were available within 21 days.
Results: The sensitivity and specificity compared to the proportion method, was 98.5% and 100% for INH, 98.3% and 96.2% for RIF, 91.7% and 100% for KAN, 78.8% and 97.3% for OFX, 100% and 100% for AMK and CAP, respectively.
Conclusion: The results lead to the conclusion that direct nitrate reductase assay, in this new format, is an accurate, quick and inexpensive method to determine the susceptibility profile of M. tuberculosis and may become an alternative for countries with limited resources.
Key words: Mycobacterium tuberculosis, tuberculosis, sputum, antitubercular agents, drug resistance, nitrate reductase.
doi: http://dx.doi.org/10.7705/biomedica.v35i2.2570
Evaluación de la prueba de la nitrato reductasa directa en microplaca para la detección rápida de tuberculosis multirresistente y extensamente resistente a fármacos
Introducción. Los informes sobre Mycobacterium tuberculosis resistente a múltiples fármacos están aumentando a nivel mundial y los laboratorios toman cada vez más conciencia sobre la necesidad de realizar pruebas de sensibilidad a fármacos. Debido al tiempo prolongado que se requiere para hacerlas, se han propuesto nuevos enfoques para la detección rápida de resistencia a los medicamentos, tales como la prueba de la nitrato reductasa.
Objetivo. En este estudio se propuso una prueba de sensibilidad a fármacos colorimétrica, rápida y directa, en un formato de microplacas utilizando medio sólido.
Materiales y métodos. La precisión del diagnóstico se evaluó mediante la comparación del método de las proporciones con la prueba de la nitrato reductasa directa en placas. Se descontaminaron muestras positivas congeladas de esputo y se procesaron mediante el método de Petroff. La suspensión ya descontaminada se utilizó para hacer la prueba de la nitrato reductasa directa en medio 7H11, utilizando 1 µg/ml de rifampicina, 0,2 µg/ml de isoniacida, 2 µg/ml de ofloxacina, 6 µg/ml de de kanamicina, 2 µg/ ml de amicacina y 10 µg/ml de capreomicina. Se analizaron 84 muestras y los resultados del 69 % de ellas estuvieron disponibles en 21 días.
Resultados. La sensibilidad y la especificidad de la prueba comparada con el método de las proporciones fueron de 98,5 y 100 % para la isoniacida, de 98,3 y 96,2 % para la rifampicina, de 91,7 y 100 para la kanamicina, de 78,8 y 97,3 % para la ofloxacina y de 100 y 100 % para la amikacina y la capremicina, respectivamente.
Conclusión. Este nuevo formato de la prueba de la nitrato reductasa directa es un método preciso, rápido y económico para determinar el perfil de sensibilidad de M. tuberculosis y puede convertirse en una alternativa para los países con recursos limitados.
Palabras clave: Mycobacterium tuberculosis, tuberculosis, esputo, antituberculosos, resistencia a medicamentos, nitrato-reductasa.
doi: http://dx.doi.org/10.7705/biomedica.v35i2.2570
Tuberculosis remains a major public health problem worldwide, especially in developing countries. Such situation is worsened by the increasing incidence of multidrug resistant tuberculosis (MDR-TB), caused by strains resistant to at least isoniazid (INH) and rifampicin (RIF), and of extensively resistant tuberculosis (XDR-TB) caused by a MDR-TB strain also resistant to fluoroquinolone and to at least one of the following antimicrobial agents: kanamycin (KAN), amikacin (AMK) and/or capreomycin (CAP). Tuberculosis caused by resistant strain requires an expensive, longer and less effective treatment (1).
Drug resistance surveillance data indicate that in 2013, approximately 480,000 people developed MDR-TB worldwide and approximately 210,000 deaths. An estimated 3.5% of new cases and 20.5% of previously treated cases have MDR-TB. Despite progress in the detection of MDR/RR-TB cases, a major diagnostic gap remains: 55% of reported tuberculosis patients estimated to have MDR-TB were not detected in 2013 (2).
By the end of 2013, 100 countries and territories reported at least one case of XDR-TB while countries like Iran, India and Italy have reported cases of pan-drug resistant tuberculosis (PDR-TB). Of 269 XDR-TB patients reported in 40 countries in the 2011 cohort overall, only 284 (22%) completed their treatment successfully and 438 (35%) patients died (2-5).
In countries with limited resources, where tuberculosis is endemic and MDR-TB is a serious and growing public health problem, the drug susceptibility test is still based on conventional methods of culture in solid medium as Löwenstein-Jensen and Middlebrook agar, which are laborious and time consuming, requiring at least 4 weeks of incubation after the primary isolation to consider a strain as susceptible or resistant (6).
The development of an alternative robust test, fast and inexpensive, that can be done directly from the sputum is extremely important and urgent for tuberculosis control programs and allows rapid detection of new resistant cases as well as monitoring tuberculosis patients with resistant strains (6).
One affordable option for rapid drug resistance detection is the nitrate reductase assay that uses colorimetric detection of nitrite as an indicator of growth. It requires no elaborate equipment, expensive substrates or reagents. Although nitrate reductase assay has been shown to be highly sensitive and specific in the detection of rifampicin and isoniazid resistance, additional studies are required to determine its performance in a target population, i.e., a population in which multidrug-resistant tuberculosis is suspected (7).
The aim of this study was to evaluate the performance of nitrate reductase assay in plates in a direct manner, and to determine the susceptibility of M. tuberculosis strains directly from sputum samples using INH, RIF, ofloxacin (OFX), KAN, CAP and AMK.
Materials and methods
Proportion method
The proportion method is still regarded as the gold standard for anti-TB drugs and was performed indirectly, with the isolated strains from sputum samples at the time of their arrival at the laboratory. The technique was performed in Löwenstein-Jensen media, following the simplified protocol described by Canetti, Rist and Grosset (8) in 15 ml Falcon tubes, with or without antibiotics. The antibiotics concentrations used were: 40 µ g/ml (Sigma-Aldrich, Germany, 7382), 0.2 µ g/ml INH (Sigma-Aldrich, Germany, 3377), 2 µ g/ml OFX (Sigma-Aldrich, Germany, 8757), 20 µ g/ml KAN (Sigma-Aldrich, Germany, 4000), 40 µ g/ml AMK (Sigma-Aldrich, Germany, 1774) and 20 µ g/ml CAP (Sigma-Aldrich, Germany, 4142).
For each strain, a bacterial suspension was made by transferring bacterial colonies to a tube containing glass beads and 2.5 ml of sterile distilled water. This suspension was equivalent to McFarland number 1 tube. Part of this suspension was diluted 1:100, and 200 µ l of this dilution were inoculated in Löwenstein-Jensen media without antibiotics while 200 µ l of undiluted suspension were inoculated into Löwenstein-Jensen media with antibiotics. The tubes were incubated at 37°C for 28 days when the first reading was taken. If there was not enough growth to allow results interpretation, a second reading was performed at 42 days of incubation.
The strain was considered resistant when the ratio between the growth in tubes containing antibiotic and the control tubes was higher than 1% with all antibiotics. The results obtained by the proportion method were used as gold standard to be compared with the results of direct nitrate reductase assay in plates (9).
Natural samples
A total of 94 frozen sputum samples (-20°C) from different patients non-previously decontaminated and with known susceptibility profile (determined by the proportion method) were selected from the collection of the Laboratório de Micobactérias, Universidade Federal do Rio Grande, Brazil. Direct nitrate reductase assay in plates was performed on 84 sputum samples, as 10 (10.6%) of them were contaminated. Among the chosen samples, we had 46 MDR-TB isolates, 6 XDR-TB, 2 INH resistant plus one drug, 14 isoniazid-monoresistant, 3 rifampicin-monoresistant and 13 pan-susceptible. The selection criterion was based on the susceptibility profile and provenience, since the target population was from low-income countries. In order to have a significant sampling, the samples were chosen mostly from Kinshasa, Democratic Republic of Congo, Africa, where the incidence of tuberculosis cases is 326/100,000 and 272 cases of MDR-TB were notified in 2013 (2).
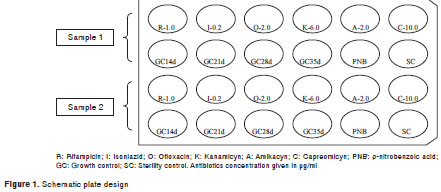
The samples were decontaminated through the Petroff modified method (10). In short, a volume of approximately 2 ml of sputum was decontaminated with an equal volume of 4% NaOH solution. Tubes containing the samples were placed in a bacteriological incubator for 15 minutes at 36±1°C for sample fluidization. After this, the tube volume was completed with sterile distilled water and the neutralizing solution (1N HCl) was dripped until an amber coloration appeared. Then, the tubes were centrifuged for 15 minutes at 3000 g , the supernatant discarded and the pellet re-suspended in 1 ml of sterile distilled water. The schematic plate design can be seen in figure 1.

Direct nitrate reductase assay in plates
The indirect method proposed by Ängeby, Klintz and Hoffner (7) was used as a model, with modifications in format, culture medium, antibiotics concentration and number of controls. A 24-well microplate was used with 2 ml of 7H11 (chosen for being a more enriched medium than Löwenstein-Jensen media, which can shorten the time for obtaining results), enriched with 10% of Middlebrook oleic acid albumin dextrose catalase enrichment, potassium nitrate (KNO 3 ) in a concentration of 1 mg/ml with or without antibiotics incorporated into each well.
Antibiotics concentrations were: 1 µ g/ml RIF (Sigma-Aldrich, Germany, 7382), 0.2 µ g/ml INH (Sigma-Aldrich, Germany, 3377), 2 µ g/ml OFX (Sigma-Aldrich, Germany, 8757), 6 µ g/ml KAN (Sigma-Aldrich, Germany, 4000), 2 µ g/ml AMK (Sigma-Aldrich, Germany, 1774), 10 µ g/ml CAP (Sigma-Aldrich, Germany, 4142) and 25 µ g/ml p-nitrobenzoic acid (PNB) (Acros Organics - A0260097). The PNB was included in this study to differentiate strains belonging to Mycobacterium tuberculosis complex from non-tuberculous mycobacterium, since the latter are the only ones that grow in the presence of this drug (11). To eliminate contamination, the cultures were observed every 2 days to check for any possible growth. The control wells were stained to confirm the presence of acid-fast growth.
After the sample decontamination and resuspension of the pellet, sterile distilled water was added to complete a volume of 3 ml. This was considered as the working solution. The working solution was inoculated into the test wells and a dilution of 1:10 was made. This last one was called the diluted solution and was inoculated into control wells.
For each sample, 200 µ l of the working solution were inoculated in test wells containing 7H11, KNO 3 , antibiotics while 200 µ l of the diluted solution were inoculated in control wells which contained only the culture medium and KNO 3 . The sterility control was done by the inoculation of 200 µ l of water in a control well.
The plate was incubated at 37°C in aerobic atmosphere, inside a sealed plastic bag. After 14 days, 0.5 ml of freshly mixed Griess reagent (1 part of 50% hydrochloric acid, 2 parts of 0.2% sulfanilamide and 2 parts of 0.1% n-1-naphthyl ethylene-ediamine) was added to control wells. If there was any color change (pale pink-strong pink), the test wells were also revealed. If there was no color change in the control well, the plate was incubated again and the procedure was repeated at 21, 28 or 35 days. A sample was considered to be resistant to a certain drug if there was any color change.
Pan-susceptible M. tuberculosis H37Rv reference strain and a known MDR M. tuberculosis isolate were used as quality controls.
Ethical aspects
This study was based on ethical principles and was approved by the Research Ethics Committee in the Area of Health of the Federal University of Rio Grande, Protocol 23116.003686/2012-31.
Results
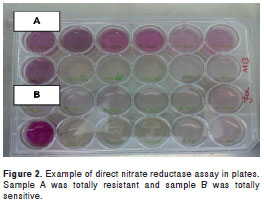
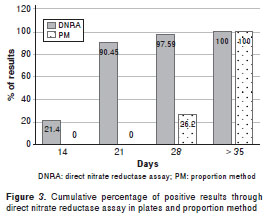
The results of 18 (21.4%), 58 (69.05%), 6 (7.14%) and 2 (2.4%) samples were obtained after 14, 21, 28 and 35, respectively, and then compared with those produced by proportion method. Direct nitrate reductase assay was easily read, one example of sensitive and resistant strains can be seen in figure 2.
Regarding the indirect proportion method, we obtained results of 22 (26.2%) of the samples in 28 days and of 62 of the samples (73.8%) in 42 days, adding to that time 21 days on average for growing strains, totaling 49 or 63 days to obtain the results of drug susceptibility testing (figure 3).
The comparison of direct nitrate reductase assay and proportion method (table 1) shows an agreement of 97.4% (491 of 504 individual sensitivity tests), 1.19% of inconclusive samples (6 of 504 tests - corresponding to 3 different samples) and 0.79% of discordant samples (4 of 504 tests). Considering the discordant samples, the sensitivity and specificity were 98.5% and 100% for INH, 98.3% and 100% for RIF, 91.7% and 100% for KAN, 81.8% and 100% for OFX, 100% and 100% for AMK and 100% and 100% for CAP. An excellent agreement was obtained between direct nitrate reductase assay and proportion method, with a kappa value of 0.96 for INH, 0.97 for RIF, 0.95 for KAN, 0.89 for OFX and 1.0 for AMK and CAP.
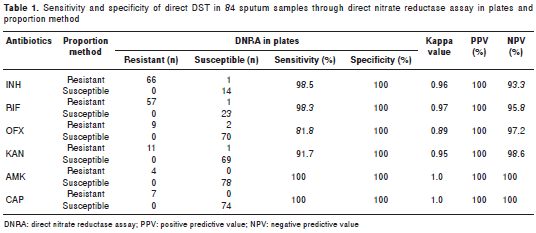
The positive predictive values were 100% for all drugs. The negative values were 93.3%, 95.8%, 97.2%, 98.6%, 100% and 100% for INH, RIF, KAN, OFX, AMK and CAP, respectively.
The accuracy of direct nitrate reductase assay in plates in relation to the gold standard was 98.8% for INH, 98.8% for RIF and KAN, 97.5% for OFX and 100% for AMK and CAP.
Discussion
In low-income countries, particularly those where the burden of tuberculosis caused by resistant strains is high, there is an urgent need for drug susceptibility testing for first and second line drugs used to treat tuberculosis. These methods should be easy to use, give rapid and accurate results and have low cost (12).
Although direct nitrate reductase assay in tube is a well-known method, the microplate format is a more compact device and permits a rapid detection of MDR-TB and XDR-TB strains in just one platform (13,14).
Furthermore, direct nitrate reductase assay reduced diagnosis time, low cost and straightfor-ward implementation (6,15) without any need for special equipment or expensive reagents and, finally, greater biosafety, since there is no need for carrying out serial dilutions, as in the proportion method (16).
In this study, approximately 69% of the results of direct nitrate reductase assay were obtained after 21 days of incubation. This represents a significant reduction of time, since the results of the proportion method performed indirectly are obtained 50 days after their arrival at the laboratory on average, since 21-28 days are required for strain isolation and 28-42 days for the drug susceptibility testing. The nitrate reductase assay is based on the detection of nitrate reduction as an indicator of growth, and the results, therefore, are obtained before any macroscopic growth can be visually detected. Despite this reduction in time, there is a need for further comparative studies with direct drug susceptibility testing.
Our results confirm previous findings with direct nitrate reductase assay carried out with INH, RIF, STR and ethambutol (EMB) (1,6,12,15-24). The sensitivity and specificity varied between 87-100%, 87.5-100%, 66.6-100%, 55-100%, respectively.
In a multicentric study carried out by Martin, et al ., the accuracy for RIF ranged between 93.7% and 100%, for INH, between 88.2% and 100%, for OFX, between 94.6% and 100%, and for KAN the accuracy was 100%. They also had the majority of positive results at 21 days (25). Imperiale, et al ., found that the direct nitrate reductase assay had a turn-around time of 16.9 days to obtain results while that of the indirect MGIT 960 system was 29 days (26).
In our study, there was a high agreement between the direct nitrate reductase assay and the proportion method for five of the six antibiotics tested. The high performance for INH and RIF must be highlighted, since these are the most commonly used drugs in tuberculosis treatment (16).
In 2005, Martin, et al ., reported the evaluation of the nitrate reductase assay for the detection of OFX resistance and found complete concordance with the proportion method. Rosales, et al ., evaluated the nitrate reductase assay for the rapid detection of resistance to OFX and KAN in Honduras. They found good specicity for both drugs, but lower sensitivity for detecting resistance to KAN (27). Visalakshi, et al ., evaluated the nitrate reductase assay for rapid detection of resistance to KAN, EMB, OFX, cycloserine and para aminosalicylic acid. They obtained a sensitivity ranging between 86.4% and 100%, while specificity ranged between 98.4% and 100% (28).
For OFX, the result of a sensitivity of 81.8% was not as good. However, the specificity remained excellent (100%). The accuracy of the method for this particular drug was 97.5%. A small number of discordant samples hampered the analysis of the test for this drug, since the number of OFX resistant samples tested was small; it is suggested that further studies are needed to correctly classify a case as MDR-TB or XDR-TB using this new method. Martin, et al., also found lower sensitivity for OFX due to the very low number of strains resistant to this drug (25).
Unlike our results, Kurup and George found a higher sensitivity for RIF than for INH, as well as greater negative agreement indices than positive ones (28).
Although direct nitrate reductase assay in microplates has significant advantages, it is important to take into account limitations such as the possibility that resistant strains may have lower metabolic activity, a fact that can affect the expression levels of enzymes responsible for nitrate reduction that cannot be detected by nitrate reductase assay (18). Another considerations refers to a possible interaction between the antibiotics and KNO 3 , which is still unknown and should be considered in future assessments (14).
The improvement in the diagnosis of susceptible and resistant tuberculosis strains through molecular methods is significant; however, these methods detect well-known resistance molecular bases, mainly to RIF and INH. The molecular bases of resistance to second-line drugs are less known, so the capacity to detect resistance to these drugs have a low level of accuracy through molecular methods while phenotypic methods such as the direct nitrate reductase assay detect resistance independently of its molecular bases and have a higher sinsitivity for the detection of tuberculosis resistant strains.
In conclusion, our results suggest that the direct nitrate reductase assay can be a useful tool for multidrug-resistant tuberculosis detection since it is fast, low-cost and accurate, and it determines both first and second-line drugs profile of susceptibility directly from the clinical sample.
Financial support
This study received financial support from the Ministério da Saúde, Brasil , and the Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq , Process number 133851/2010-9.
Conflict of interests
No significant conflicts of interests exist with any of the companies or organizations whose products or services are mentioned in this article.
Corresponding author:
Pedro Eduardo Almeida da Silva, Laboratório de Micobactérias, Faculdade de Medicina, Universidade Federal do Rio Grande, CEP 96200-400, Rio Grande, RS, Brasil Phone number: (55 53) 3233 0314; fax: (55 53) 3233 8895 pedrefurg@gmail.com
1. Affolabi D, Odoun M, Martín A, Palomino JC, Anagonou S, Portaels F. Evaluation of direct detection of Mycobacterium tuberculosis rifampin resistance by a nitrate reductase assay applied to sputum samples in Cotonou, Benin. J Clin Microbiol. 2007;45:2123-5. http://dx.doi.org/10.1128/JCM.00691-07
2. World Health Organization . Global Tuberculosis Report 2014. Fecha de consulta: 13 de diciembre de 2014. Disponible en: http://www.who.int/tb/publications/global_report/en/.
3. Migliori GB, De Iaco G; Besozzi G, Centis R, Cirillo DM. First tuberculosis cases in Italy resistant to all tested drugs. Euro Surveillance. 2007;12:3194.
4. Udwadia ZF, Amale RA, Ajbani, KK, Rodrigues C. Totally drug-resistant tuberculosis in India. Clin Infec Dis. 2012;54:579-81. http://dx.doi.org/10.1093/cid/cir889
5. Velayati AA, Masjedi MR, Farnia P, Tabarsi P, Ghanavi J, Ziazarifi AH, et al. Emergence of new forms of totally drug-resistant tuberculosis bacilli – super extensively drug-resistant tuberculosis or totally drug-resistant strains in Iran. Chest. 2009;136:420-5. http://dx.doi.org/10.1378/chest.08-2427
6. Montoro E, Lemus D, Echemendia M, Martín A, Portaels F, Palomino JC. Comparative evaluation of the nitrate reduction assay, the MTT test and the resazurin microtitre assay for drug susceptibility testing of clinical isolates of Mycobacterium tuberculosis . J Antimicrob Chemother. 2005;55:500-5. http://dx.doi.org/10.1093/jac/dki023
7. Ängeby KA, Klintz L, Hoffner SE. Rapid and inexpensive drug susceptibility testing of Mycobacterium tuberculosis with a nitrate reductase assay. J Clin Microbiol. 2002;40:553-5. http://dx.doi.org/10.1128/JCM.40.2.553-555.2002
8. Canetti G, Fox W, Khomenko A, Mahler HT, Menon NK, Mitchison DA, et al. Advances in techniques of testing mycobacterial drug sensitivity, and the use of sensitivity tests in tuberculosis control programmes. Bull World Health Organ. 1969; 41 :21-43.
9. Canetti G, Rist N, Grosset J. Mesure de la sensibilité du bacille tuberculeux aux drogues antibacillaires par la methode des proportions. Ver Tuberc et Pneum. 1963;23:217-72.
10. Organización Panamericana de la Salud. Manual de bacteriología de la TBC: técnicas y procedimientos básicos. Washington, D.C.: OPS; 1973.
11. Barreto AM, Caldas PC, Santos LM, Martins FM, Medeiros RF. Avaliação do ácido p-nitrobenzóico para identificação do complexo Mycobacterium tuberculosis no sistema MB/BacT. Boletim de Pneumologia Sanitária. 2006;14:17-9.
12. Affolabi D, Odoun M, Sanoussi N, Martín A, Palomino C, Kestens L, et al . Rapid and inexpensive detection of multidrug resistant tuberculosis with the nitrate reductase assay using liquid medium and applied directly to sputum samples. J Clin Microbiol. 2008;46:3243-5. http://dx.doi.org/10.1128/JCM.00083-08
13. Coban AY, Birinci A, Ekinci B, Durupinar B. Drug susceptibility testing of Mycobacterium tuberculosis with nitrate reductase assay. Int J Antimicrob Agents. 2004;24:106-08. http://dx.doi.org/10.1016/j.ijantimicag.2004.02.027
14. Martín A, Montoro E, Lemus D, Simboli N, Morcillo N, Velasco M, et al . Multicenter evaluation of the nitrate reductase assay for drug resistance detection of Mycobacterium tuberculosis . J Microbiol Methods. 2005;63:145-50. http://dx.doi.org/10.1016/j.mimet.2005.03.004
15. Solis LA, Shin SS, Han LL, Llanos F, Stowell M, Sloutsky A. Validation of a rapid method for detection of M. tuberculosis resistance to isoniazid and rifampin in Lima, Perú. Int J Tuberc Lung Dis. 2005;9:760-4.
16. Shikama ML, Silva, RF, Villela G, Sato DN, Martins MC, Giampaglia CM S, et al . Multicenter study of nitrate reductase assay for rapid detection of rifampicin-resistant M. tuberculosis . Int J Tuberc Lung Dis. 2009;13:377-80.
17. Visalakshi P, Meharwal SK, Myneedu VP, Behera D. Evaluation of direct method of drug susceptibility testing of Mycobacterium tuberculosis to rifampicin and isoniazid by nitrate reductase assay in a national reference laboratory. Diagn Microbiol Infect Dis. 2010;66:148-52. http://dx.doi.org/10.1016/j.diagmicrobio.2009.09.008
18. Ani AE, Dalyop YB, Agbaji O, Idoko J. Drug susceptibility test of Mycobacterium tuberculosis by nitrate reductase assay. J Infect Dev Ctries. 2009;3:16-9. http://dx.doi.org/10.3855/jidc.100
19. Gupta A, Anupurba S. Direct drug susceptibility testing of Mycobacterium tuberculosis against primary anti-TB drugs in northern India. J Infect Dev Ctries. 2010;4:695-703. http://dx.doi.org/10.3855/jidc.1079
20. Gupta M, Singh NP, Kaur IR. Evaluation of nitrate reductase assay for direct detection of drug resistance in Mycobacterium tuberculosis : Rapid and inexpensive method for low-resource settings. Indian J Med Microbiol. 2010;28:363-5. http://dx.doi.org/10.4103/0255-0857.71816
21. Kumar M, Khan IA, Verma V, Kalyan N, Qazi GN. Rapid, inexpensive MIC determination of Mycobacterium tuberculosis isolates by using microplate nitrate reductase assay. Diagn Microbiol Infect Dis. 2005;53:121-4. http://dx.doi.org/10.1016/j.diagmicrobio.2005.05.011.
22. Mandal PK, Basnyat S, Khadka DK, Bhatta DR. Evaluation of nitrate reductase assay (NRA) for rapid detection of drug resistant tuberculosis of national tuberculosis center, Nepal. SAARC Journal of Tuberculosis, Lung Diseases and HIV/AIDS. 2010;7:26-30. http://dx.doi.org/10.3126/saarctb.v7i1.3960
23. Martín A, Panaiotov S, Portaels F, Hoffner S, Palomino JC, Ängeby K. The nitrate reductase assay for the rapid detection of isoniazid and rifampicin resistance in Mycobacterium tuberculosis : A systematic review and meta-analysis. J Antimicrob Chemother. 2008;62:56-64. http://dx.doi.org/10.1093/jac/dkn139
24. Musa HR, Ambroggi M, Souto A, Ängeby KA. Drug susceptibility testing of Mycobacterium tuberculosis by a nitrate reductase assay applied directly on microscopy-positive sputum samples. J Clin Microbiol. 2005;43:3159-61. http://dx.doi.org/10.1128/JCM.43.7.3159-3161.2005
25. Martín A, Imperiale B, Ravolonandriana P, Coban AY, Akgunes A, Ikram A, et al . Prospective multicentre evaluation of the direct nitrate reductase assay for the rapid detection of extensively drug-resistant tuberculosis. J Antimicrob Chemother. 2014;69:441-4. http://dx.doi.org/10.1093/jac/dkt353
26. Imperiale BR, Morcillo NS, Palomino JC, Vandamme P, Martín AJ. Predictive value of direct nitrate reductase assay and its clinical performance in the detection of multi- and extensively drug-resistant tuberculosis. Med Microbiol. 2014;63:522-7. http://dx.doi.org/10.1099/jmm.0.070219-0
27. Martín A, Palomino JC, Portaels F. Rapid detection of ofloxacin resistance in Mycobacterium tuberculosis by two low-cost colorimetric methods: Resazurin and nitrate reductase assays. J Clin Microbiol. 2005;43:1612-6. http://dx.doi.org/10.1128/JCM.43.4.1612–1616.2005
28. Visalakshi P, Arora J, Behera D. Evaluation of nitrate reductase assay for rapid detection of resistance to second-line drugs in Mycobacterium tuberculosis in a tertiary care hospital. Diagn Microbiol Infect Dis. 2010;68:236-40. http://dx.doi.org/10.1016/j.diagmicrobio.2010.06.017