ARTÍCULO ORIGINAL
doi: http://dx.doi.org/10.7705/biomedica.v33i0.686
1 Grupo de Medicina Tropical, Instituto Colombiano de Medicina Tropical-Universidad CES, Medellín, Colombia
2 Grupo Centauro, Facultad de Ciencias Agrarias, Universidad de Antioquia, Medellín, Colombia
Authors ´ contributions:
Andrés F. Londoño participated in field data collection.
Victoria E. Murillo analyzed histopathological results.
Juan D. Rodas and Piedad Agudelo-Flórez were the principal researchers of the initial project, conducted the analysis of results, and wrote, edited and proofread the article.
Recibido: 12/04/12; aceptado: 23/11/12
Introduction: Histopathological changes by Leptospira in naturally infected rodent reservoirs have been poorly described.
Objective: The aim of the current study is to describe renal histopathology associated with leptospirosis infection of naturally infected rodents captured in the urban area of the city of Medellin, Colombia.
Materials and methods: We performed hematoxilin-eosin (H-E) on kidney samples collected from 254 captured rodents. The positive samples were processed by Warthin Starry (W-S) staining and PCR- LipL 32.
Results: Fifty one rodent kidneys showed H-E histopathological changes that consisted of inflammatory infiltrate with lympho-plasmocitary cells and histiocytes. We performed W-S staining and PCR- LipL 32 to 67 kidney samples, including the 51 that had shown detectable changes by H-E and 16 (8%) of 203 rodents with negative results. Eight of the samples that tested positive for H-E (15.7%) were also positive for W-S staining. All negative for H-E were also negative for W-S staining. Of the W-S positive samples also tested for culture only three tested positive for both. Additionally, 47 (92.1%) samples positive for H-E were positive for PCR; while eleven of the 16 (68.8%) negative for H-E were positive for PCR. The samples positive for PCR were subsequently tested for culture and 11 (23.4%) were positive. Seven samples were positive for PCR and W-S and three were positive for PCR, W-S and culture. All of the PCR- LipL 32 fragments were sequenced and showed specific amplicons for L. interrogans .
Conclusions: The Leptospira infection was confirmed in all of the animals tested. The only histological kidney lesion attributable to leptospiral infection in the reservoir was interstitial nephritis.
Key words: Leptospira /pathology, hematoxylin, polymerase chain reaction, rodent.
http://dx.doi.org/10.7705/biomedica.v33i0.686
Alteraciones histopatológicas en ratas naturalmente infectadas con Leptospira
Introducción. Los hallazgos histopatológicos ocasionados por Leptospira spp. han sido poco estudiados en poblaciones de roedores naturalmente infectados.
Objetivo. Describir la histopatología renal asociada con las infecciones naturalmente adquiridas en un grupo de roedores capturados en el área urbana de Medellín, Colombia.
Materiales y métodos. Se llevaron a cabo coloraciones de hematoxilina y eosina de los riñones de 254 roedores recolectados en el área de estudio. Las muestras positivas se procesaron con la coloración de Warthin-Starry y mediante reacción en cadena de la polimerasa (PCR)-LipL32.
Results. Se observaron cambios histopatológicos con hematoxilina y eosina en 51 riñones de roedores, que consistieron en infiltrado inflamatorio con linfoplasmocitos e histiocitos. Se utilizó coloración de Warthin-Starry y PCR-LipL32 en 67 muestras de riñón que incluyeron las 51 muestras que tuvieron cambios detectables por hematoxilina y eosina y 16 de 203 (8 %) muestras con resultados negativos. Ocho de las muestras positivas por hematoxilina y eosina (15,7 %) también fueron positivas por la coloración de Warthin-Starry. Las muestras negativas por hematoxilina y eosina (8 %) también fueron negativas con la coloración de Warthin-Starry. Tres de las ocho muestras positivas por esta última, también lo fueron por cultivo. Además, 47 (92,1 %) muestras positivas por hematoxilina y eosina fueron positivas por PCR. Del grupo de 16 negativos por hematoxilina y eosina, 11 (68,8 %) fueron positivos por PCR. De las muestras positivas por PCR, 11 también lo fueron por cultivo (23,4 %). Siete muestras fueron positivas por PCR y Warthin-Starry y tres lo fueron por PCR, Warthin-Starry y cultivo. Todos los fragmentos de la PCR-LipL32 fueron secuenciados y mostraron secuencias específicas de L. interrogans .
Conclusiones. Se confirmó la infección por Leptospira y la única lesión presente en el reservorio atribuible fue la nefritis intersticial.
Palabras clave: Leptospira /patología, hematoxilina, reacción en cadena de la polimerasa, roedores.
http://dx.doi.org/10.7705/biomedica.v33i0.686
Leptospirosis is caused by pathogenic spirochaetes of the genus Leptospira (1). In 1913, the organism that acts as the causal agent of leptospirosis was identified and it was found that rats are a reservoir for human transmission (2). Several Leptospira spp have been isolated from a wide range of animal reservoir species such as cows, pigs, dogs, horses and others such as bats and field and synanthropic rodents (1). Infection can be transmitted among animals by direct contact with infected urine, venereal route and mucosa; open wounds; indirect exposure through water, vegetation or soil; or food contaminated with urine of mammalian hosts (1, 3). This zoonotic disease has emerged as an epidemic in urban slums where inadequate sanitation has produced ideal conditions for rat-borne transmission (1, 4).
Infection is initially expressed as a leptospiremic phase, in which the bacteria spreads itself rapidly through bloodstream and can be found throughout the body, especially in the kidneys, liver, lungs, adrenal glands and meninges; it can also reach fetal membranes and reproductive tract. Next is a subsequent leptospiruric phase in which specific antibodies counteract blood bacteria, which survive mainly by colonizing the kidney where they form micro colonies in renal tubules and are subsequently excreted through urine for an indeterminable amount of time (1, 3, 4).
In the case of adapted animals and reservoirs, these bacteriuria periods may last throughout the animal´s life. It is well known that rodents are the main reservoir for Leptospira because they are persistent renal carriers (1). Transmission of leptospirosis requires continuous enzootic circulation of the pathogen among animal reservoirs. Leptospira serovars demonstrate specific, although not entirely exclusive, host preferences. For example, rats ( Rattus rattus and R. norvegicus ) serve as reservoirs for the Icterohaemorragiae serogroup, whereas house mice ( Mus musculus ) are the reservoirs for the Ballum serogroup. Furthermore, serovars often do not cause serious disease in hosts to which they are highly adapted (1, 3, 5).
Knowledge regarding disease presentation or lack of infection in naturally resistant hosts allows understanding of important factors of leptospirosis such as physiology and immunopathology from indigenous or native isolates, though these aspects have been scarcely explored in Colombia. In a study previously performed in the country, Leptospira interrogans serovars Pomona and Icterohaemorrhagiae were isolated from the kidneys of Rattus norvegicus captured in Pomona infected piggeries. Eight Leptospira isolations were carried out from 111 cultured kidneys. Seven isolates corresponded to serovar Icterohaemorrhagiae; one corresponded to Pomona. Kidney tubular degeneration, focal mononuclear infiltrations and fibroplasia were the most common morphological traces remaining (48.6% of the rat kidneys) after the leptospirosis attack (6).
The current study aims to describe renal histopathology associated with leptospirosis infection of naturally infected rodents captured in the urban area of Medellin, Colombia. The results permit us to describe the histopathological changes caused by Leptospira spp in an animal test subject´s renal tissue for asymptomatic leptospirosis obtained through natural infection and associate this with the presence or absence of the microorganism on rodent kidneys determined by culture or Warthin Starry (W-S). Likewise, the presence of the microorganisms in the kidneys of rodents is described through silver staining and PCR- LipL 32 against the pathogenic Leptospira spp and confirmed by sequencing the amplified PCR products.
Materials and methods
Study site description
Rodents were captured in the urban farmer´s market Plaza Minorista Jose Maria Villa in Medellin, Colombia. Trapping was conducted throughout 28 sessions during 7 months between August 2006 and April 2007. Rodents´ capture, anesthesia, euthanasia, processing of tissue samples and follow up tests are previously described (7).
Renal histopathology
One of the kidneys from each of the captured rats was fixed in neutral-buffered formalin and embedded in paraffin. Tissue sections (2-4 µm) were stained with hematoxilin-eosin (H-E) and examined using whole slide imaging. All kidneys displayed morphological alterations by H-E and 8% of the kidneys without changes were processed by Warthin-Starry (W-S) stain. The lesions were classified according to severity levels as light (<30% affected), moderate (30-50%) and severe (>50%). We performed W-S staining on 67 kidney samples, including 51 that displayed detectable changes by H-E and sixteen (8%) of 203 rodents showing negative results.
Kidney culture for detection of Leptospira
One half of the second kidney was macerated in 3 ml of physiological saline solution in a sealed plastic bag. Fifty milliliters of each macerated tissue sample was cultured in Fletcher semisolid medium followed by incubation at 28 to 30 °C. The cultures were examined under a dark-field microscope once per week during 4 months for positive or negative status (7). The other half of the second kidney was preserved at -70 °C for PCR Leptospira studies.
Leptospira detection on kidney by PCR
In order to determine the presence of pathogenic Leptospira all kidneys with morphological alterations evident by H-E and 8% of the kidneys without changes were processed by PCR- LipL 32 (7). One half of the sample tissue was used for genomic DNA extraction using QIAmp DNA Mini Kits (QIAGEN, Hilden, Germany) according to the manufacturer´s instructions. This DNA was used in a PCR reaction for pathogenic serovars using primers LipL 32/270F (8). To confirm that the amplicons were Leptospira sequences, all PCR -LipL32 products were purified (Microcon® Centrifugal Filter Devices, Billerica, MA, USA) and sequenced in both directions on a 3130 DNA sequencer (Applied Biosystems, Foster City, CA, USA). Sequences were examined using the Basic Local Alignment Search Tool (BLAST) to confirm the presence of Leptospira spp in the sample.
Analysis of results
The results of each rodent were registered in a format which included a rodent code, capture date, macroscopic description, sample histopathology of H-E and the presence or absence of Leptospira of W-S, PCR and culture. A descriptive statistical analysis of the data yielded mainly proportions, rates, and frequencies. Additionally, Chi² tests were performed to establish the association between each of the variables studied and the presence of changes of H-E in captured rodents. Data were analyzed using the statistical program EPI INFO 2003 (Redmond, WA, USA).
Results
We collected 254 Rattus norvegicus rodents . Of the total animals captured, 156 (61%) were male, 179 (71%) were adults and 234 (92%) appeared healthy during clinical examination.
Histopathological findings
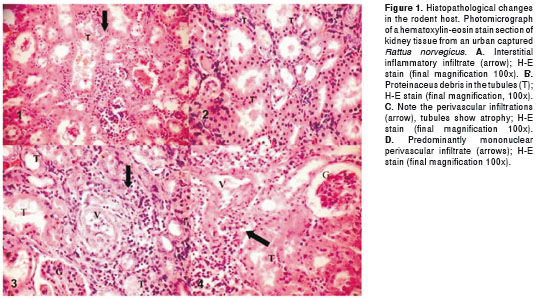
The average kidney size was approximately 1 to 1.5 cm, and none of the examined kidneys showed evident macroscopic lesions; however, during the examination with H-E staining, kidneys from 51 rodents (20.1%) exhibited histopathological changes. The most common lesion was interstitial nephritis that consisted of inflammatory infiltrate with predominance of lympho-plasmocitary cells and histiocytes that varied among light (39.2%), moderate (47.1%) and severe (13.7%) (Figure 1A). In severe cases we observed focal compromise, particularly of the perivascular location, most commonly affecting cortical and small to medium-sized vessels. Seventeen (33.3%) exhibited changes such as tubular atrophy with dilatation, thyroidization and hyaline cylinders in the tubule interiors. Glomerular changes in three kidneys (5.8%) consisted of increased cellularity; no kidneys exhibited glomerular sclerosis, mesangioproliferative glomerulonephritis or interstitial fibrosis (Figure 1B-1D).

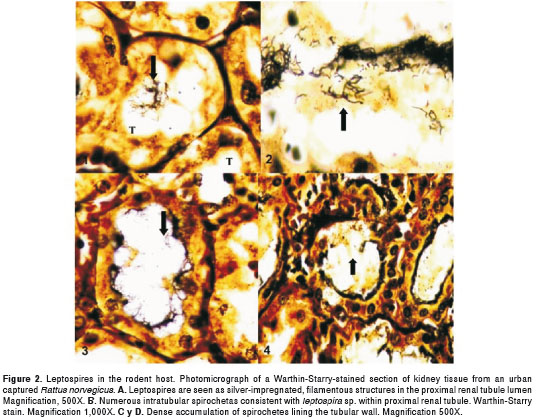
Sixty-seven kidney samples were stained with W-S, including the 51 that had shown detectable changes by H-E and 16 (8%) of 203 rodents with negative results. Within the H-E positive group, eight (15.7%) also tested positive for W-S staining. Among these we observed dense aggregates of microorganisms similar to Leptospira in the lumen of the tubules convoluted and attached to the luminal surface (Figures 2A-2D). We did not find bacteria at the interstitial level, invading the epithelial cells or in the lumen of blood vessels. None of the samples that resulted in negative H-E exhibited positive W-S staining.
Leptospiral findings
Out of sixty-seven kidneys analyzed by H-E stain, 26 (38.8%) were positive for culture. All isolates showed typical morphology and characteristic motility of the genus Leptospira through direct examination under the dark-field microscope. Out of the 51 rodents that showed histopathological changes by H-E, 19 (37.2%), had positive culture with severe grade interstitial nephritis being the most prominent feature. Interestingly enough, 7 (43.8%) of the 16 H-E negative rodents (corresponding to the 8% processed) had positive culture.
Additionally, when PCR- LipL 32 was tested 47 samples positive by H-E were also positive by PCR (92.1%); while five of 16 that were found negative by H-E, (31.3%), tested by PCR were also confirmed negative. The samples positive by PCR were tested by culture and 11 (23.4%) were positive, but only seven samples were positive by PCR and W-S; six were positive by PCR, W-S and culture. All of the PCR- LipL 32 fragments were sequenced and showed specific products for L. interrogans confirming the presence of pathogenic Leptospira in these samples.
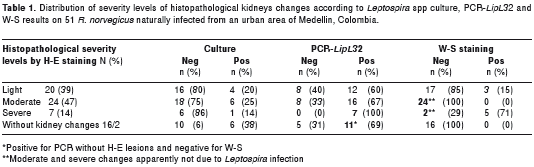
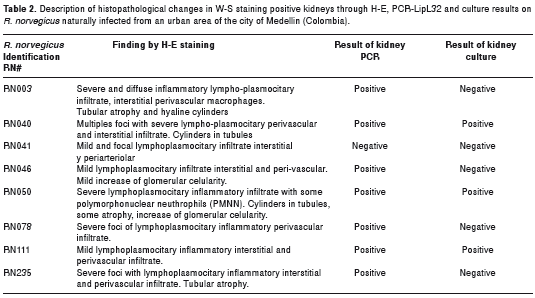
The distribution of severity levels of histopathological changes according to Leptospira spp culture, PCR- LipL 32 and W-S status are described in Table 1. Additionally, the specific description of histopathological changes observed by H-E, PCR- LipL 32 and culture obtained to samples positive by W-S, is shown in table 2.
We did not find statistically significant association between the independent variables of sex, age and health and the dependent variables, histopathological changes and positive culture (p> 0.05) by logistic regression.
Discussion
The current study describes the renal histopathology related with rodents naturally infected with leptospirosis hunted from the urban area of Medellin, Colombia. A broad range of morphological alterations were detected in the kidneys from captured rats but interstitial nephritis was the only morphological alteration seemingly or partially attributable to leptospiral infection observed in all W-S and PCR- LipL 32 positive rodents.
In the persistent colonization cases as in reservoir animals, leptospires cause systemic infection but are subsequently cleared from all organs except the renal tubules. Colonized tubules are densely populated with leptospires, which aggregate into an amorphous, biofilm-like structure. Rats have been shown to excrete leptospires in high concentrations (10 7 organisms per ml for up to 9 months) after experimental infection (4). The renal tubule is an immune-privileged site, a feature that may contribute to high-grade persistence of the pathogen. Moreover, the leptospires shed in the urine down-regulate the expression of proteins that are recognized by the humoral immune response in rats (4).
Previous studies evaluating renal morphology in captured urban rats carrying leptospires have all reported that interstitial nephritis was the principal change attributed to leptospires (9-11). The pathogenesis of naturally-occurring leptospiral infection in rats has not been extensively studied and is complicated by a wide array of potentially confusing environmental factors that may also induce pathology in wild urban rats (11). The present study evaluated renal pathology in urban rats from a city where the prevalence of leptospiral isolation by kidney culture was approximately 20% (7). The finding of interstitial nephritis, however, cannot be attributed solely to leptospires since these rodents could also be exposed to other unknown environmental infectious agents that could cause interstitial nephritis (12). This could be implied from table 1 where some of the moderated histopathological changes (8/24) are negative even to PCR- LipL 32 and also because Leptospira spp was detected by culture and PCR in kidneys without evident H-E changes.
Even though we did not obtain statistically significant association between histopathological changes in W-S staining positive kidneys through H-E, PCR- LipL 32 and culture results on R. norvegicus (Table 2) and although these are naturally infected rats from an urban area, we still believe that under the scant body of evidence about the effect of leptospiral infection upon its natural reservoirs these findings are worth publishing. This matter is biologically significant and deserves further study.
In seven of eight rodent kidneys positive for W-S we identified the LipL32 as a virulence factor. This lipoprotein is highly conserved among pathogenic Leptospira spp, whereas there are no orthologues in the saprophytic L. biflexa . The role of this major outer membrane protein in pathogenesis remains unclear (4), but it should be noted that LipL 32 was highly recognized in the W-S positive kidneys in our survey showing a possible role in the chronic renal carrier state associated with Leptospira spp. The knowledge of mechanisms leading to chronic renal shedding and the information on the persistence of leptospires adapted host remains poorly understood. Both host and microbiological factors probably contribute to the severity of leptospiral infection (10).
The confirmation obtained in this study by means of histopathological and molecular methods and its connection with previous Colombian reports of rodents trapped in piggeries (6) and general urban prevalence reports of human leptospirosis, 11% in Medellin City (13), 23.3% in the Cauca Valley (14) and 12.5% in the region of Urabá (15) underlines the plausible importance of leptospirosis for public health in Colombia. Human outbreaks in 1995 (16) related to rainy seasons, floods and domestic rat infestation, with a mortality rate higher than 17%, and an animal outbreak among susceptible Cebidae ( 17) support the role of rats as important reservoirs for pathogenic leptospires that might cause human leptospirosis in Colombia. However, case control studies are required to identify the real risks associated with rodents and the leptospirosis in the human population.
Additionally, we studied the dissemination of Leptospira in infected rodents by kidney cultures, but this method has low sensitivity. In the present study, we combined different approaches, including PCR, to gain a better understanding of the disease pathology. The PCR- LipL 32 used to evaluate the presence and dissemination of L. interrogans in a carrier model of leptospirosis indicates that this assay provides an accurate method for determining leptospires in kidneys displaying a ranging severity of histopathological changes and also is applicable to direct sequencing for determining Leptospira s pp and will help in the design of further infection experiments.
None
This work was supported by the Comité para el Desarrollo de la Investigación (CODI EO 1206), Universidad de Antioquia , and the Dirección de Investigación, Universidad CES , Colombia.
Correspondencia: Lorenzo Cáceres, Instituto Conmemorativo Gorgas de Estudios de la Salud, Apartado Postal 0816-02593, Panamá, República de Panamá. Teléfono (507) 527 4963; fax (507) 527 4889 lcaceres@gorgas.gob.pa y cacereslorenzo@gmail.comcacereslorenzo@gmail.com
1. Bharti AR, Nally JE, Ricaldi JN, Matthias MA, Díaz MM, Lovett MA, et al . Leptospirosis: A zoonotic disease of global importance. Lancet Infect Dis. 2003;3:757-71. http://dx.doi.org/10.1016/S1473-3099(03)00830-2
2. Inada R, Ido Y, Hoki R, Kakeno R, Ito H. The etiology, mode of infection and specific therapy of Weil´s disease ( Spirochaetosis icterohaemorrhagica ). J Exp Med. 1916; 23:377-403.
3. Levett PN. Leptospirosis. Clin Microbiol Rev. 2001;14:296-326. http://dx.doi.org/10.1128/CMR.14.2.296-326.2001
4. Ko AI, Goarant C, Picardeau M . Leptospira : The dawn of the molecular genetics era for an emerging zoonotic pathogen. Nat Rev Microbiol. 2009;7:736-47. http://dx.doi.org/10.1038/nrmicro2208
5. Thiermann AB . The Norway rat as a selective chronic carrier of Leptospira icterohaemorrhagiae. J Wildl Dis. 1981;17:39-43.
6. Morales GA, Guzmán VH, Beltrán LE . Leptospirosis in Colombia: Isolation of Leptospira spp. from the kidneys of brown rats ( Rattus norvegicus ) trapped on infected piggeries. Trop Anim Health Prod. 1978; 10 : 121-3. http://dx.doi.org/10.1007/BF02235323
7. Agudelo-Flórez P, Londoño AF, Quiroz VH, Ángel JC, Moreno N, Loaiza ET, et al . Prevalence of Leptospira spp. in urban rodents from a groceries trade center of Medellin, Colombia. Am J Trop Med Hyg. 2009;81:906-10. http://dx.doi.org/10.4269/ajtmh.2009.09-0195
8. Levett PN, Morey RE, Galloway RL, Turner DE, Steigerwalt AG, Mayer LW. Detection of pathogenic leptospires by real-time quantitative PCR. J Med Microbiol. 2005;54:45-9.
9. Sterling C R, Thiermann AB. Urban rats as chronic carriers of leptospirosis: An ultrastructural investigation. Vet Pathol. 1981;18:628-37.
10. Tucunduva de Faria M, Athanazio DA, Gonçalves Ramos EA, Silva EF, Reis MG, Ko AI . Morphological alterations in the kidney of rats with natural and experimental Leptospira infection. J Comp Pathol. 2007;137:231-8. http://dx.doi.org/10.1016/j.jcpa.2007.08.001
11. Athanazio DA, Silva EF, Santos CS, Rocha GM, Vannier-Santos MA, McBride AJ, et al. Rattus norvegicus as a model for persistent renal colonization by pathogenic Leptospira interrogans . Acta Trop . 2008;105:176-80. http://dx.doi.org/10.1016/j.actatropica.2007.10.012
12. Winter H, Watt D. Klossiella hydromyos n. sp. from the kidneys of an Australian water rat ( Hydromys chrysogaster). Vet Pathol. 1971;8:222-31. http://dx.doi.org/10.1177/030098587100800304
13. Sebek Z, Sixl W, Valova M, Marth E, Dock M, Reinthaler FF. Serological investigations for leptospirosis in humans in Columbia. Geogr Med Suppl. 1989;3:51-60.
14. Ferro BE, Rodríguez AL, Pérez M, Travi BL . Seroprevalencia de infección con Leptospira en habitantes de barrios periféricos de Cali, Colombia. Biomédica. 2006;26:250-7.
15. Agudelo-Flórez P, Restrepo BN, Arboleda M . Situación de la leptospirosis en el Urabá antioqueño colombiano: estudio seroepidemiológico y factores de riesgo en población general urbana. Cad Saude Pública. 2007;23:2094-102. http://dx.doi.org/10.1590/S0102-311X2007000900017
16. Epsein PR, Calix O, Blanco J . Climate and disease in Colombia. Lancet. 1995;346:1243-4. http://dx.doi.org/10.1016/S0140-6736(95)91856-6
17. Szonyi B, Agudelo-Flórez P, Ramírez M, Moreno N, Ko A. An outbreak of severe leptospirosis in capuchin (Cebus) monkeys. Vet J. 2011; 188:237-39. http://dx.doi.org/10.1016/j.tvjl.2010.05.002